Haptic feedback solutions for TAVI procedures
Aortic stenosis is a common valve disease, associated with a high rate of death among untreated patients (50% in the first two years after symptoms appear). It is characterised by a narrowing of the aortic valve opening, usually induced by age-related progressive calcifications. The most common treatment is open-heart surgical aortic valve replacement (SVR), although it is not recommended for at least 30% of the patients, due to their advanced age and multiple coexisting conditions, which make the surgical procedure too risky. Transcatheter aortic valve replacement (TAVR) was introduced in the last decade as a minimally invasive alternative, and consists in implanting inside the diseased valve a bioprosthetic substitute, delivered on a catheter through either the femoral artery or the ventricular apex. Improved prostheses and delivery systems, as well as the increased experience of interventionalists, has favoured the reduction of complication rates and contributed to the evolution of TAVR from an experimental strategy to a routine intervention.
Nevertheless, TAVR is still affected by major procedural complications, including:
- Neurological events, caused by dislodged calcific material due to catheter manipulation;
- Aortic regurgitation, often associated with prosthetic valve malpositioning;
- Renal failure, induced by the large amount of contrast agent injected during the implantation to correctly deliver the device.
These problems are related to the poor manoeuvrability of current delivery systems and the inadequacy of site information, provided to the clinician during the whole procedure.
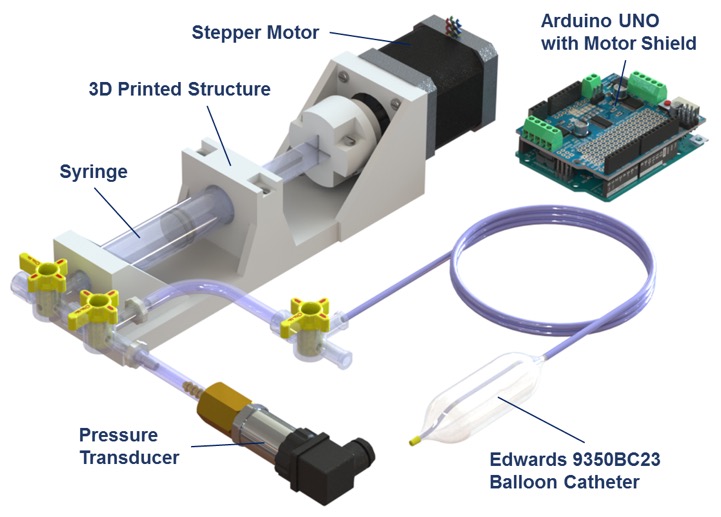
The aim of this project is to integrate the latest developments in robotics and sensing in a novel delivery platform, capable of intravascularly sensing and reacting to key site information (catheter-vessel interaction forces, prosthetic valve orientation, distance from critical areas) and providing guidance to the surgeon through haptic feedback. The main objectives are:
- embedded microscale sensing technologies in the catheter ;
- adopt state-of-the-art actuation methods for endovascular navigation and relevant control techniques;
- develop a haptic feedback interface;
- validate our approach through in vitro experiments.
 Close
Close







